Electrical Engineering Intern
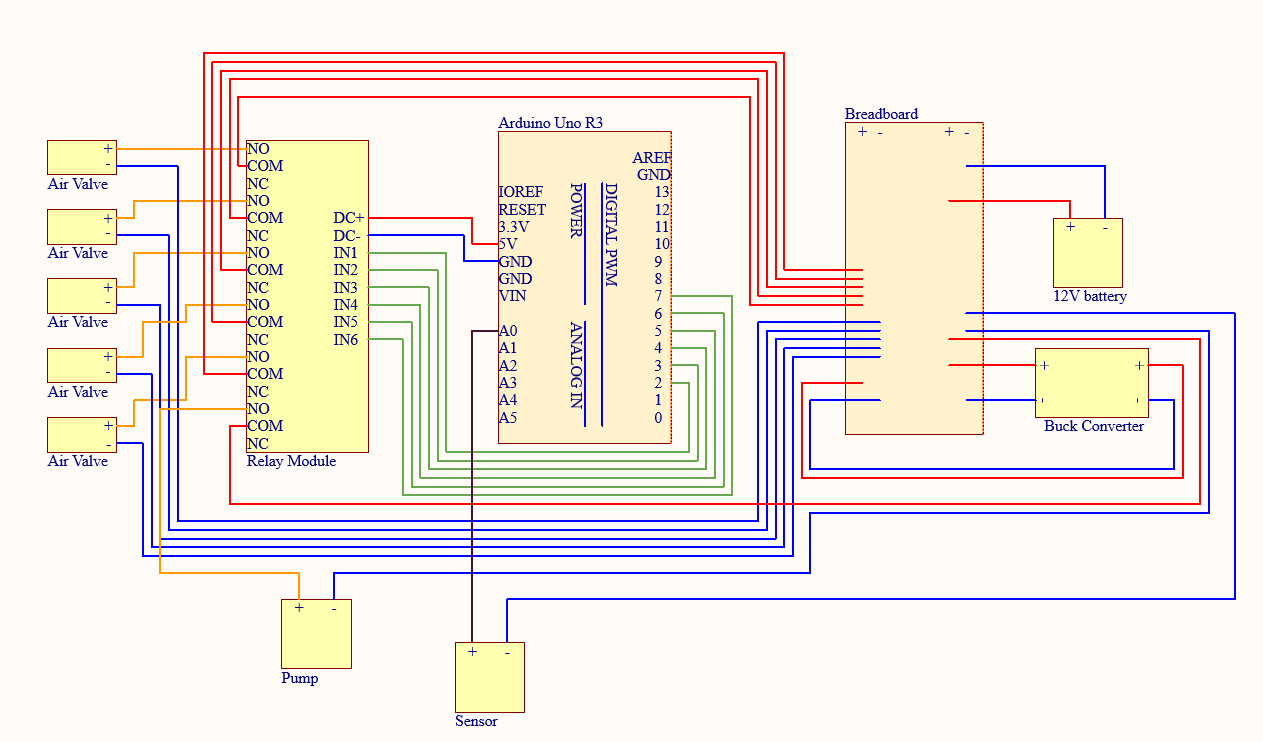
Contributed across two summer internships to the fabrication and development of autonomous unloading robots. Specialized in intricate PCB soldering, IPC software loading, and full electrical system assembly. Helped design and build quality-control test hardware, authored detailed build documentation, retrofitted prototype robots to test new technology, and partnered directly with the CEO to deliver live customer and investor demos.
Highlights
- Designed and built quality control test fixtures
- Authored step-by-step assembly and quality control guides used by the manufacturing and operations teams
- Retrofitted prototype robots to test new improvements
- Supported CEO in customer and investor demos